AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
 To do this, the amount of buffer used in the transfer is limited to what is contained in the transfer sandwich. Transfer speed is improved over wet tank by maximizing the current passing through the gel instead of around the gel. In a semi-dry protein transfer, the transfer sandwich is placed horizontally between two plate electrodes. Semi-dry electroblotting (Semi-dry transfer) With increasing time, however, there is a risk of over-transfer (stripping, blow through) of the proteins through the membrane, especially for lower molecular weight (<30 kDa) proteins when using membranes with a larger pore size (0.45 µm). The transfer efficiency improves with increased transfer time and is better, in general, for lower molecular weight proteins than higher molecular weight proteins. Transfer efficiencies of 80–100% are achievable for proteins between 14–116 kDa. A high field option exists for transferring a single gel, which may bring transfer time down to as little as 30 minutes, but it requires the use of high voltage (up to 200 V) or high current (up to 1.6 A) and a cooling system to dissipate the tremendous heat produced. Multiple gels may be electrotransferred in the standard field option, which is performed either at constant current (0.1 to 1 A) or voltage (5 to 30 V) from as little as 1 hour to overnight. The supported gel sandwich is placed vertically in a tank between stainless steel/platinum wire electrodes and the tank is filled with transfer buffer. The gel is then placed in the “transfer sandwich” (filter paper-gel-membrane-filter paper), cushioned by pads and pressed together by a support grid. 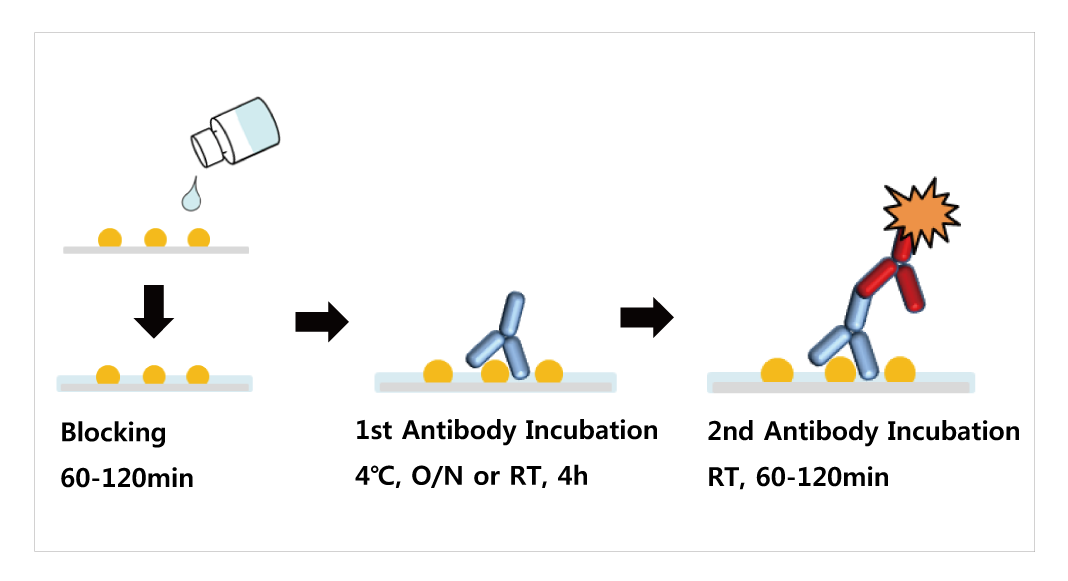
When performing a wet transfer, the gel is first equilibrated in transfer buffer. The efficiency of protein transfer can be affected by the chemistry, thickness of the gel, the molecular weight of the proteins being transferred, the type of membrane and transfer buffers used, and the transfer method. In addition to the challenges of immunodetection in the protein blotting workflow, the transfer of proteins from a gel matrix to a membrane is a potential hurdle. An appropriate method is then used to detect the localized probe to document the location and relative abundance of the target protein. The transferred protein is then probed sequentially with antibodies and detection probe (e.g., enzyme, fluorophore, isotope). Better target protein accessibility on the membrane by macromolecules like antibodiesĪfter transfer, the membrane must be blocked to prevent non-specific binding of the antibody to the membrane surface.Better handling capability offered by the membrane compared to a fragile gel.Protein transfer from gel to membrane is necessary for two reasons: 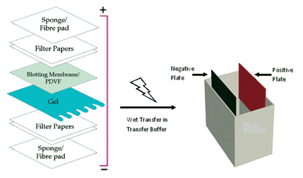
In procedures where protein separation is not required, the sample may be directly applied to the membrane by spotting using an approach called dot blotting. After electrophoresis, the separated proteins are transferred, or "blotted", onto a solid support matrix, usually a nitrocellulose or polyvinylidene difluoride (PVDF) membrane. The transfer buffer was glycine and tris with 20% methanol, in distilled water.The first step in a western blotting procedure is to separate the proteins in a sample by size using denaturing gel electrophoresis (i.e., sodium dodecyl sulfate polyacrylamide gel electrophoresis or SDS-PAGE) or native PAGE. The only thing that I notably did differently was that I made the transfer buffer up the day before and left it in the ice machine overnight. I'm not really sure what went wrong here to be honest. Upon dis-assembling the transfer module I noticed that the sponged inside all had turned yellow, however only the sponge touching the positive electrode, and only the side of the sponge facing the positive electrode. After 2h the blot was still only at 4v, and had barely transferred at all. 
I checked the tank and the power pack temperature, and both were not noticeably hot, so I don't think the pack was struggling with high resistance. However in this case after 1hr the blot was still only running at 4v. Normally when transferring two blots in the same tank we run for 2hrs at 20v, as the tank takes about 30 minutes to get up to 20v. I have a western blot troubleshooting question that I haven't been able to find the answer to in manufacturer troubleshooting guides.Īs a bit of background, I was transferring 2 western blots together in the same tank (we use a BioRad tank that fits 2 transfer modules side-by-side). 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
